Description: BOPA Film for Pharmaceutical Packaging POA Pharmaceutical Model
ULTRA NY POA Pharmaceutical Model is a highly efficient, medical grade nylon film developed exclusively for the global pharmaceutical industry. The two-step biaxial orienting process employed to create BOPA Film for Pharmaceutical Packaging produces a product that provides a desirable balance of mechanical strength and excellent levels of puncture resistance. Pharmaceutical Blister Films are most commonly used in cold-forming blisters and tropical blister packaging within the pharmaceutical industry.
The toughness and anti-twist characteristics of BOPA Film for Pharmaceutical Packaging allow it to greatly minimise the breakage and/or delamination of the film throughout the high-speed stamping process. Additionally, the BOPA Film for Pharmaceutical Packaging maintains a uniform structural integrity which allows it to function effectively across a wide range of temperatures; in addition to this, BOPA Film for Pharmaceutical Packaging functions as an oxygen barrier film for medicines allowing for extremely low oxygen transmission rates (OTR) of approximately 40cc/(·day · atms) for the heavier versions of the film thereby eliminating oxidation of active ingredients; BOPA Film for Pharmaceutical Packaging is additionally a highly transparent and shiny film, making it possible for users to clearly identify capsules and tablets.
BOPA Film for Pharmaceutical Packaging may be supplied either with a single side or with both sides corona-treated for optimal surface energy for laminations to aluminium foil or PVC.
Additionally, the high annual production capacity of 150,000 tons of BOPA Film for Pharmaceutical Packaging ensures a consistent supply of the film for the global pharmaceutical composite bag market. BOPA Film for Pharmaceutical Packaging is utilised as a strong and reliable barrier layer in the three-side seal and four-side seal bags for biologics and enzyme preparation. The ability of BOPA Film for Pharmaceutical Packaging to provide the physical support required for the sterile medical device blister packaging caps will enable pharmaceutical companies to utilise a Cold Form Blister Packaging Film that protects pharmaceuticals from damage caused by moisture or heat degradation in hot and humid tropical environments.
Benefits of BOPA Film for Pharmaceutical Packaging POA Pharmaceutical Model
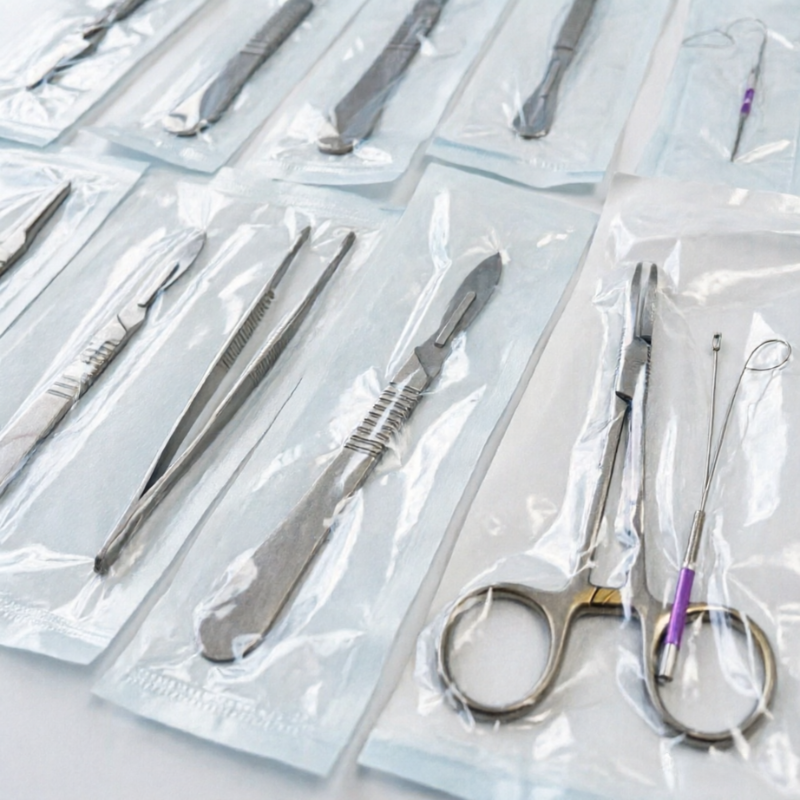
a. High-stamping quality reliability of our nylon film is achievable through its ability to support extreme pressures within a cold-forming process without cracking at the edge of blister packaging.
b. Robust physical protection is offered through the puncture-resistance of the material, supporting forces of 10 N to 20 N and protecting tablets and sharp medical devices from damaging forces.
c. Exceptional gas-barrier properties of the material provide extended shelf life of very reactive pharmaceutical compounds.
d. Superior adhesion of the interlayers of the material is achieved as a result of Corona Treatment on one or both sides of the material, resulting in good bonding of Medical Grade Nylon Film with aluminium foil and PVC.
e. Clear and easy-to-spot identification and clarity can be achieved using the high transparency and low haze (as low as 4.5%) of the material, enabling easy visual inspections of medicines.
f. High resistance to mechanical damage prevents wear and tear of laminated products, including business cards.
| Thickness μm | Corona | Width mm |
| 15/20/25/30 | One Side or Both Sides | 300-1500 |
Basic Info.
| Model NO. | ULTRANY® | Thickness | 15/20/25/30μm |
| Transport Package | Carton Package | Specification | POA Pharmaceutical Model |
| Trademark | Changsu | Origin | China or Indonesia |
ULTRANY® POA Pharmaceutical Model – Application (BOPA Film for Pharmaceutical Packaging):
Liquid Vitamin Sachets: Protecting volatile nutrients in liquid food supplements using the Oxygen Barrier Film for Medicine technology.
Bone-in Meat Packaging: Preventing punctures from sharp bone fragments using the High Puncture Resistant Blister Film.
High-End Tea Pouches: Locking in delicate aromas and preventing moisture ingress using the Medical Grade Nylon Film as a barrier layer.
Vacuum-Packed Seafood: Maintaining seal integrity during deep freeze storage using the toughness of Pharmaceutical Blister Film.
ULTRANY® POA Pharmaceutical Model BOPA Film for Pharmaceutical Packaging can also be utilised for other Applications:
Pharmaceutical Cold Forming Blisters: The most common application of BOPA Film for Pharmaceutical Packaging is to protect tablets and capsules.
Medical Device Caps: Using Cold Form Blister Packaging Film for sterile catheters, scalpel blades, and implantable devices.
Biologics Composite Bags: Providing an oxygen barrier film for medicine, probiotics and enzyme preparation sensitive to light.
Heavy Industrial Liners: Using the 30µm High Puncture Resistant Blister Film for large size medical supplies or sharp tools.
BOPA Film for Pharmaceutical Packaging POA Pharmaceutical Model Product Parameters
| Typical Properties | Test Method | Units | Typical Value | |
| Thickness | - | μm | 12 | |
| Average Thickness Tolerance | ISO4593 | % | ±3.0 | |
| Tensile Strength | MD | ASTM D882 | MPa | 220 |
| TD | 240 | |||
| Elogation at Break | MD | ASTM D882 | % | 80 |
| TD | 120 | |||
| Heat Shrinkage | MD | ASTM D1204 (160ºC, 5min) | % | ≤ 2.0 |
| TD | ≤ 2.0 | |||
| Dynamic COF | Static/Kinect | ASTM D1894 | - | 0.55/0.40 |
| Surface Tension | Treated Side | ASTM D2578 | mN/m | 54 |
| Untreated Side | 39 | |||
| Puncture Resistance | GB/T 10004-2008 | N | 7.5 | |
| Haze | ASTM D1003 | % | 4.0 | |
| Oxygen Transmission Rate | 23℃, 50%RH | cc/㎡.day.atm | 70 | |
Length (m): Standard 4000 ~ 24000
With(mm): 300-1500
Paper core (inches): 3, 6
Packaging: 9, 4 or 2 rolls/pallet
Remark:
Data in the above section is released to provide a comprehensive and reliable reference of normal film behaviour. Data in the above section should only be referenced for comparison purposes.
As we continue to innovate, Changsu reserves the right to change product characteristics at any time without prior notice.
Got Questions for ULTRANY® POA Pharmaceutical Model BOPA Film for Pharmaceutical Packaging? We've Got Answers!
How does this film perform during high-speed cold stamping?
BOPA Film for Pharmaceutical Packaging is specifically designed for high-speed stamping with excellent deep-drawing performance. This Pharmaceutical Blister Film features high puncture resistance and balanced mechanical strength, which effectively reduces the break or delamination in different directions during the stamping process.
Why is BOPA Film for Pharmaceutical Packaging ideal for tropical regions?
This Cold Form Blister Packaging Film is specifically designed for pharmaceuticals requiring storage in high-temperature, high-humidity climates. The Medical Grade Nylon Film effectively resists humid and hot environments, preventing pharmaceuticals from becoming damp or oxidising, which significantly extends the overall product shelf life.
What are the barrier properties of this Pharmaceutical Blister Film?
BOPA Film for Pharmaceutical Packaging acts as a reliable Oxygen Barrier Film for Medicine. For example, the 25μm and 30μm versions provide an Oxygen Transmission Rate of 40 cc/(·day·atm), which safeguards sensitive biologics and powders from oxidative deterioration during long-term storage.
Can this film be used for sharp medical instruments?
Yes, BOPA Film for Pharmaceutical Packaging is a High Puncture Resistant Blister Film with a resistance of up to 20N for the 30μm model. This Medical Grade Nylon Film protects sterile packaging for catheters and scalpels from sharp edges, ensuring the integrity of the sterile barrier system.
Does BOPA Film for Pharmaceutical Packaging support complex lamination structures?
Absolutely, as BOPA Film for Pharmaceutical Packaging offers one or both sides corona treatment with surface tension as high as 54 mN/m. This ensures that the Medical Grade Nylon Film forms a strong bond with aluminium foil or Tyvek in multi-layer Cold Form Blister Packaging Film structures.
Service Advantages
1
24/7 Technical Support
Our expert team is available around the clock to assist with optimising your cold forming and high-speed stamping parameters.
2
Customized Configurations
We provide professional length, width, and core size options to match your specific production line requirements.
3
Stable Global Supply
A massive 150,000-ton yearly capacity ensures that pharmaceutical companies have a dependable and consistent supply of medical-grade films.
Certificates

FSSC 22000

ISO 22000:2018

MSDS

ROHS

ISO 15987:2003

ISO 14001:2015

ISO9001

IATF 16949:2016
Introduction of the Company
Xiamen Changsu Industrial Co., Ltd. (Changsu) was established in 2009 and is a world-class advanced film materials technology enterprise that integrates R&D, intelligent manufacturing, and international applications. Changsu has a total area of over 450,000 sq. ft. of production space in both China and Indonesia, and is recognised around the world as a leading provider of high-performance films.
The mission of Changsu is "Better Materials, Better Life," and it will focus on developing sustainable and functional films for improving human health and safety through its work on new energy films, bio-based biodegradable films, and functional BOPA films. The development and sale of these products are ranked at the top globally.
In addition, Changsu has had the honour of being the first Chinese member of the UN FAO SAVE FOOD initiative, where we have contributed our material innovations for enhancing global food safety. Through continued innovation in the field of material technology, Changsu is committed to creating a safer, healthier and more sustainable future for all people.
Want to know more? Contact us today for pricing information, sample availability, or to obtain a custom quotation.
+86-592-6892968






